Paschen Calculator
Louis Carl Heinrich Friedrich Paschen, born in 1865 in Schwerin, Germany, made significant contributions to physics, most notably formulating Paschen's Law during his doctoral research in 1889. This law reveals that the voltage needed for an electrical spark across a gap between electrodes depends on the gas pressure and gap distance, a principle with profound physical implications. Overcoming challenges, including displacement by the Nazis, Paschen held professorships at several German universities, including Leibniz University Hannover and the University of Berlin, until his death in 1947. His enduring legacy in physics is evidenced by ongoing use of his research, particularly in understanding gas behavior.




(Bild: Max-Planck-Gesellschaft, Archiv, Berlin)
History
What is the Paschen Law?
Paschen's Law is derived based on the interplay between electric field strength, gas pressure, and gap length. Mathematically, the law is formulated by expressing the ionization coefficient (α) as a function of electric field (E) and gas pressure (p). The core equation (α/p= f(E/p)) links α, the number of ionizations per unit path length, with E and p.
In essence, Paschen's Law states that V is a function of the product pd, indicating that the breakdown voltage is determined by both the gas type (through the ionization coefficient). The resulting curve, known as the Paschen Curve, plots V against pd, revealing a minimum breakdown voltage at a specific pd value, which is characteristic of each gas.
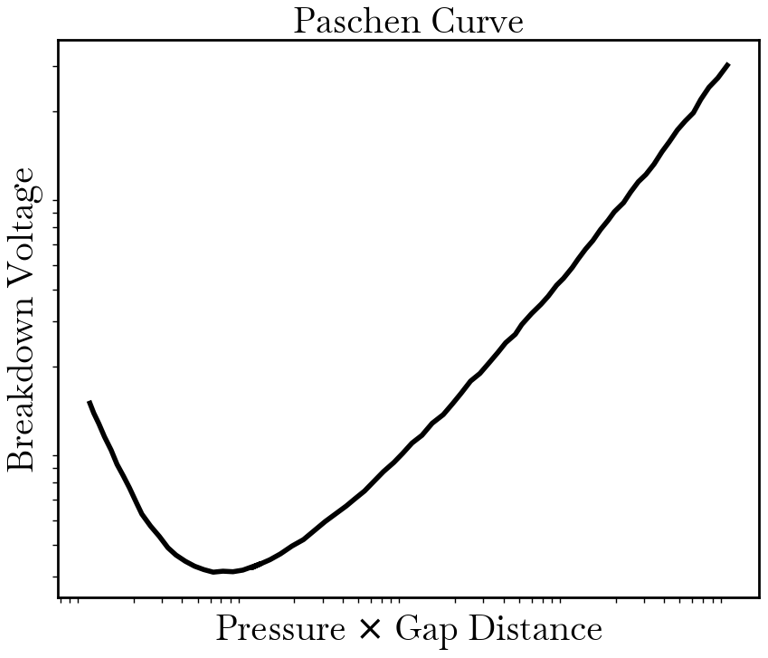
Key Considerations
Electric Field Uniformity: The law assumes a uniform electric field and may not apply to needle-plane configurations.
Gap Distance: The Law fails for very small electrode gaps, where field emission leads to lower-than-expected breakdown voltages.
Gas Composition: It is formulated for pure gases and might not hold for gas mixtures or contaminated gases.
Secondary Effects: The law does not account for the effects of electrode material and surface conditions.
Paschen Calculator
References
[1] aa
